|
9/10/2023 0 Comments Primary protein structure 
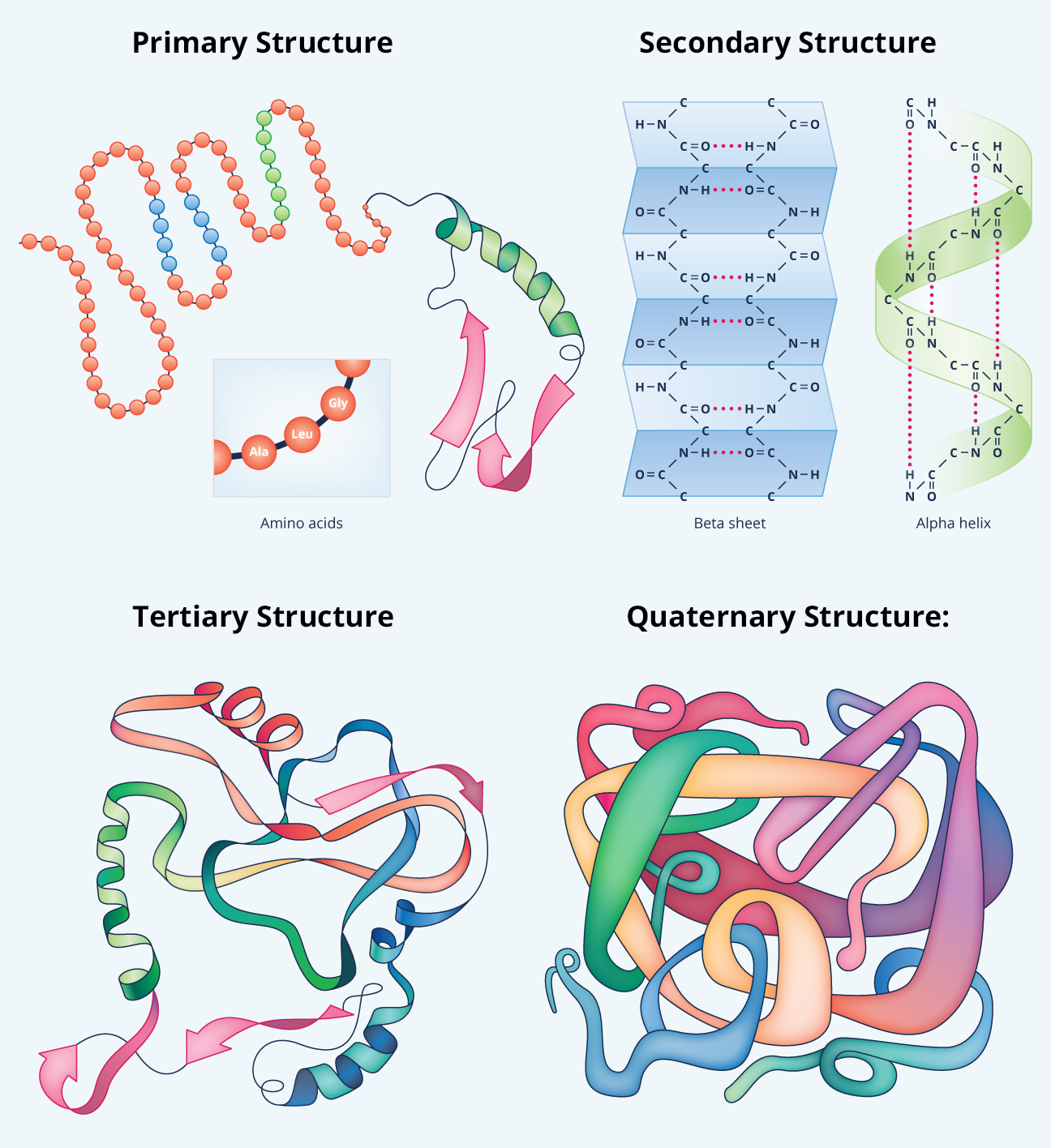
Hydrostatic bonds - form between the hydroxyl (OH) group and an adjacent hydrogen molecule, providing a strong bond between polar R groups.Fig 3 - Structure of an alpha helix and beta-pleated sheet Tertiary Protein Structure The sheets can be in parallel or antiparallel. Beta-pleated sheet - formed by hydrogen bonds between the carboxyl group of one amino acid on one sheet and the hydrogen molecule of an amino acid on another sheet.The strong bonds and stability of this structure give it a strong tensile strength, which allows it to form the shape seen in DNA. Alpha-helix - a coil formed by hydrogen bonds between the carbonyl group and the amino group of different amino acids.Primary Protein StructureĪny mutation in this amino acid sequence can affect protein folding, leading to problems with the protein's function. 12 of these can be synthesised in the body, while the other 9 must be consumed in the diet and are termed essential amino acids. There are a total of 21 amino acid types based on their different R groups. The R group gives the amino acid specific features according to its polarity and charge, which then affect the chemical and biological properties of the protein. Their structure consists of three main groups as seen in figure 1, namely the amino group or N terminus, the carboxyl group or C terminus, and the R group which contains the functional component of the amino acid. Amino acidsĪmino acids are the basic building blocks of proteins. In this article, we will discuss the structure and function of proteins, and consider their clinical relevance. Depending on the structure of the protein they will have different functions within the body, including structural, regulatory, contractile and protective roles. Proteins are made up of amino acids which undergo various stages of folding to form their shape and structure. Examples of fully functional proteins include hormones like insulin, haemoglobin, enzymes and intracellular signalling structures. It is formed via bonds between the R groups of different amino acids within the polypeptide chains, which help to give the protein its shape.Īt this point, the protein is fully functional and able to perform its specific role(s) within the body. Quaternary protein structure is when multiple polypeptide chains link together to form a functioning unit. Hydrophobic bonds – form between non-polar groups and commonly involve the benzene group.They usually occur between two cysteine amino acids, which contain sulphur within their R groups. Covalent disulphide bonds – form between sulphide groups within the R group of amino acids. /protein-structure-373563_final11-5c81967f46e0fb00012c667d.png)
They can be disrupted by the presence of other charged molecules near them.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
